



On December 22nd, the Food and Drug Administration (FDA) approved Novo Nordisk’s oral semaglutide tablet, a 25 mg dose of the same active ingredient found in injectable Wegovy and Ozempic. Marketed under the brand name Wegovy, the tablet...

On November 25, 2025, the Centers for Medicare & Medicaid Services (CMS) released negotiated maximum fair prices (MFPs) for 15 high-cost Medicare Part D drugs under the Inflation Reduction Act, effective January 1, 2027. The drugs selected...

There are many benefits to moving from Retiree Drug Subsidy (RDS) to an Employer Group Waiver Plan (EGWP):
✔️ Higher subsidies – EGWPs can provide more money in subsidies, as they can receive both...

For 2026, the maximum out of pocket (OOP) cost sharing limit for the Affordable Care Act (ACA) has been increased to $10,600 for individual/self-only coverage and $21,200 for family/non-self-only coverage. This reflects an increase from 2025...
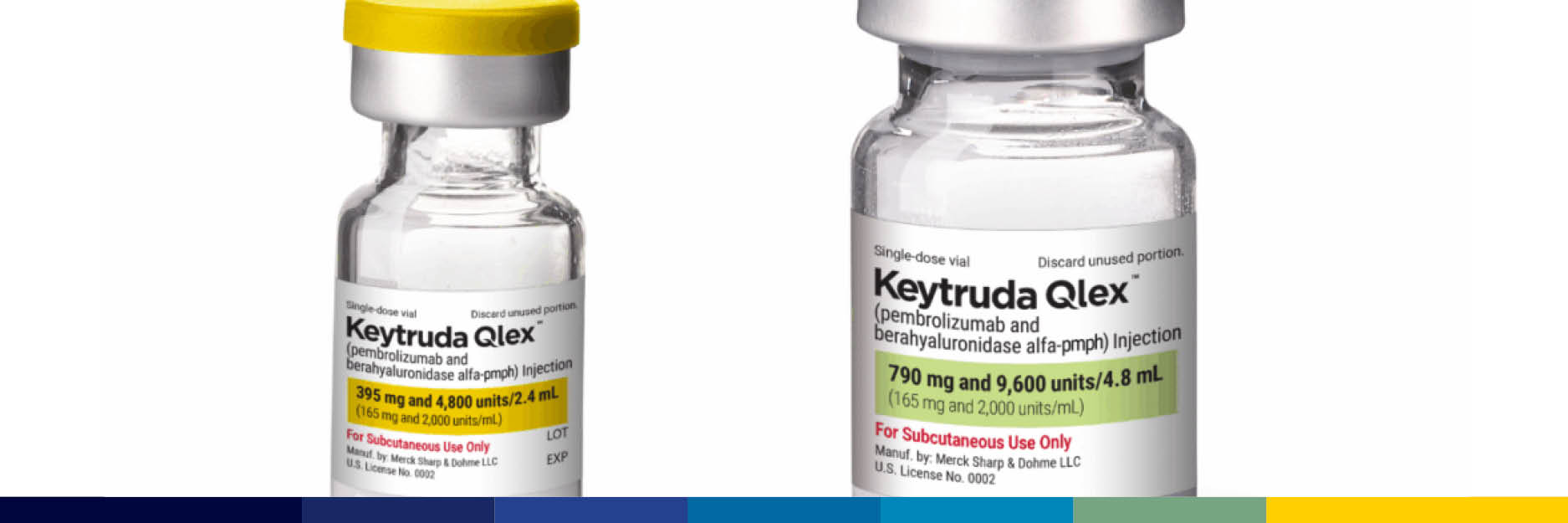
As several companies advance in developing biosimilars for Keytruda, Merck & Co. has introduced Keytruda Qlex – a subcutaneous formulation of their leading cancer therapy. Industry analysts suggest this innovative launch is strategically positioned to...





